
Buprestidae of South Australia Introduction
About the beetles
The Buprestidae (or jewel beetles) constitute just one of many beetle families – but it is a large and diverse one – the eighth largest in the world (Bouchard et al. 2017). Although the family has a cosmopolitan distribution, nearly all of the buprestid species found in Australia, and a large proportion of the genera, are unique to this continent, consistent with its long history of isolation. Australia has over 1300 described Buprestidae species (ABRS 2020), representing 5–6% of its total known beetle fauna drawn from 121 families (Yeates et al. 2003). A notable feature of our buprestid fauna is the massive radiation of species in tribe Stigmoderini. This tribe includes the largest Australian genus, Castiarina, as well as the biggest beetles, Temognatha, and appears to have evolved together with plants of the myrtle family (Myrtaceae) that produce abundant flows of nectar, including genera such as Eucalyptus, Leptospermum, Melaleuca and Hysterobaeckea.

South Australia (SA) has a surprisingly large and diverse buprestid fauna, but there are relatively few species endemic to this State alone. Although lacking centres of very high species-richness like those found in south-west Western Australia and the Sydney region of New South Wales, the central position of SA enables a good representation of the more widely occurring southern species from both Western Australia and the eastern states, and its extensive arid zone makes a further contribution. The current count of SA buprestid species is over 340.

Castiarina grata feeding on Leptospermum flower (R)
Flower-feeding buprestid beetles are generally brightly coloured and readily encountered, although their clustering behaviour makes them patchily distributed. On the other hand, the leaf-feeding species are often more cryptic and detecting them usually requires careful observation or sweeping foliage with insect nets. Buprestids generally contain the bitter compound buprestin, and their bright colours and patterning are used to advantage in deterring predators. This is evident from the prevalence of mimicry series, where buprestid species that are not closely related share colouration patterns and closely resemble one another or toxic beetles of other families.


Sex pheromones are produced by many insects to enable mates to be located by scent, but none have been detected in Buprestids. Instead, it appears that Buprestids, with their large eyes and varied colourations, find their mates visually using colour cues. This is supported by results for colour lures reported on this website which show a tendency for attraction to colours characteristic of the beetles themselves. Also, the attraction of many flower-feeding species to white or pale pink lures, and some leaf-feeding ones to bright green lures, suggests that colour is also used in locating food resources.

Most buprestid larvae are wood-borers, although unlike many longicorn beetle larvae, they are generally not considered to be troublesome pests in SA. Some larvae feed on the wood of stems, some in roots; some prefer live wood, some dead wood; a few have very different hosts. Members of the genus Ethonion, for example, are gall-makers in bush-peas, while Paracephala, Synechocera, and Meliboeithon are known to develop in the stems of grasses, stems of sedges, and rhizomes of sedges, respectively.

The wood-boring habit of most species makes study of their life cycles more difficult. Nevertheless, many new breeding records have been discovered in this project, and it is planned to progressively display relevant images and data on the website as results get formally published. These breeding records reveal a high degree of specificity in the plants used as larval hosts, at plant family, genus or even species level. Many are understorey shrubs and smaller plants with woody bases (as opposed to the larger Eucalypts and Acacias that are well recognised as hosts for some species). This explains why substantial areas of bushland with intact understories are the best places to find buprestid beetles. Conservation of buprestid beetles requires the retention and management of bushland areas and their protection from too frequent or extensive burning.
It should be noted that, along with other biota, jewel beetles are protected on crown land in SA. Field work and collection of voucher specimens in government reserves for this project was done under SA Scientific Research Permits issued by the SA Department of Environment and Water. The use of lures to investigate colour attraction, and most of the sampling for sub-adult stages, has occurred on private land with landholder permission.
Interpreting the data

The backbone of the data set informing these pages comprises over 6380 Buprestidae specimens that I have collected at over 1200 locations in SA, together with donated specimens (and a few temporary loans for processing) from other collectors. All together this amounts to more than 8330 specimens (and 12550 sighted individuals) at over 1670 distinct locations.
Further data has been sourced from selected specimens in museum and private collections, providing supplementary records for another 6650 or more individuals.
iNaturalist records used in this website are designated in the distribution map and
in image captions by the prefix ‘iNat-’ followed by their unique iNaturalist number
which can be used to access the original source records at
https://www.inaturalist.org/observations/ –
just copy the numerical code component and paste it on the end of the URL.
Alternatively, view the set of SA jewel beetle observations at
https://www.inaturalist.org/projects/jewel-beetles-of-south-australia?tab=observations&subtab=table.
Similarly, museum records and images obtained from the Ozcam website are referenced by
their institution catalogue numbers and can be accessed at https://ozcam.ala.org.au/search.
Some images derived from those sites have been used here under Creative Commons (CC)
licensing and further details about the licence conditions listed can be obtained
from https://creativecommons.org/about/cclicenses/.
My career as a professional botanist, entailing extensive fieldwork across SA, has equipped me with a comprehensive knowledge of the State’s vascular plant flora. As a result nearly all of my plant-associated buprestid collections have the adult or larval hosts identified to species level. This applies also to plant-associated collections contributed to this project by Alex Stolarski (in excess of 1007), as most of these were accompanied by pressed plant voucher samples.
The supplementary museum and private collections also provided some plant host details. These have been critically evaluated and amended as necessary in accordance with current taxonomy and nomenclature, or specific field knowledge of plant distributions and occurrences.
The counts of individuals presented here for host plant associations (both adult and larval), position in or on host, and even those displayed on the adult activity charts, all need to be interpreted with caution.
Firstly, it is worth remembering that there are sampling biases in regard to geography, host species, and time of year that even large museum collections accumulated over a long period by diverse collectors may not eliminate. The contribution of museum records varies greatly for the Buprestidae species presented here: some have had all or most of their records databased, some have none.
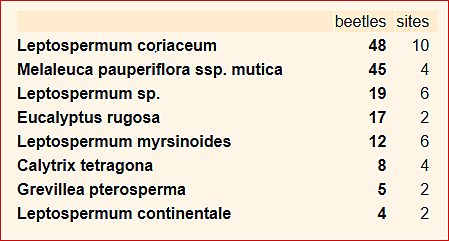
Secondly, not too much weight should be given to sequence detail in the descending sort of counts; its main purpose is to highlight those categories for which there is the strongest evidence of an association.
Thirdly, a useful general principle to apply is to weight positive data over negative data: ‘the absence of evidence is not (necessarily) evidence of absence’. That said, for host plant relationships, an absence or a low count becomes quite significant if the same host plant has very high counts for other Buprestidae species, since this means that the plant is likely to have been well-searched for Buprestidae in general. This information can be accessed readily via the ‘Host plants’ pages.
Fourthly, single records or very low counts on a plant may be quite incidental and not reflect a host preference at all; but when the overall sample size is low it is hard to tell. Consideration of the number of sites (based on 10 km grid cells) and number of regions represented (presented alongside the host plant counts) can provide extra evidence in establishing repeated patterns of host plant usage.

Non-emerged adult Anilara subcostata (R)
The different developing stages recorded, documented and tallied in the ‘Larval host plant’ section provide various strands and levels of evidence for breeding records. The various adult stage categories provide a conclusive identity, the only exception being dead adults if the remains are in poor condition. However, for pupae and larvae, evidence of their identity may be conclusive where there are subsequent reared stages, but less certain where it relies merely on repeated association with adult stages and/or host plants. In a few cases larvae have been matched to adults using DNA barcodes and this is noted in the text.
Note that the distribution maps exclude records of low geographical resolution (greater than 50 km), and thus many important historical collections given simply as ‘Mount Lofty Ranges’, ‘Kangaroo Island’ or ‘River Murray’ are omitted from the maps even if they are the only record for a region.
The website is still a work in progress, and it is planned to add more images, particularly for those species without specimen images, and to include many more text notes. Note that compiled details for some species, particularly breeding records and images, will be initially withheld and progressively released to allow for their publication in the scientific literature first.
Acknowledgements
I have greatly enjoyed collaborating with Alex (A.M.P.) Stolarski who has contributed enormously to this project. Alex collected many of the more recent specimens, and, through his keen observation and insights, is the discoverer of many new breeding records. His collections are not distinguished by their own voucher code but are included here under the PL cataloguing identifiers.
My interest in buprestids was first aroused by the publication in 2006 of Shelley Barker’s monumental treatise ‘Castiarina: Australia’s richest jewel beetle genus’. It was also greatly encouraged by D. (Andy) Young who suggested a DNA sequencing project and then seeded it with a gift of tissue samples and associated specimens representing 48 species that he collected from Kangaroo Island (indicated in images by the voucher number prefix ‘DAY’). Barcode sequencing and analysis was undertaken with Mark Stevens of the SA Museum, eventually for over 790 buprestid specimens from more than 170 species. The results support the species groups established within Castiarina by (Barker 2006) and shed light on relationships between species, informing the notes for some of the species in these web pages.

Allen Sundholm has provided much support and information over the years. I much appreciate the way he has freely shared his extensive knowledge of the family. Allen joined me on several early field trips, including a most memorable one in which we ‘rediscovered’ Temognatha congener, a species that had not been reported for over 40 years in SA. I have also benefited from information, insights, and images shared by Mark Hanlon, David Knowles, Magnus Peterson and Michael Powell.
I am grateful to the successive collection managers Jan Forrest, Peter Hudson and Matthew Shaw for access to the SA Museum (SAMA) collection. I also thank Peter Hudson and Alex Stolarski for their work in setting up trials on private land to investigate the attraction of buprestids to coloured lures.
I acknowledge with thanks the permissions granted for use of selected SA records from the following sources: the private collections of Allen Sundholm, Mark Hura, Richard Glatz, and Bryan Haywood; vouchered image records on iNaturalist of Ethan Beaver, Naomi Betts, Geoff Cox, Matt Endacott, Ned Fisher, Ralph Foster, Mark Hura, Anthony Paul and Andrew Thornhill; early access to collections of Remko Leijs being submitted to SAMA. I also thank Dan Duval (SA Seed Conservation Centre, Adelaide Botanic Gardens), Denzel Murfet and Loraine Jansen for generously making their plant images available for the ‘Host plant’ pages.
Finally, these web pages would never have been produced if not for the generosity and extraordinary abilities of Rex. Rex developed this website, writing the programming required to process and present information extracted from my databases and display it together with the images. He also suggested the ‘Host plant’ pages, came up with much of their structure, and compiled the plant images for them.
Dedication
I never cease to be astounded by the beauty and intricate functionality of the natural world. This website is dedicated in praise of the triune God, who I believe created all of nature, sustains it, and will redeem it in the promised new creation.

References cited
ABRS (2020). Buprestidae. Australian Faunal Directory. Australian Biological Resources Study, Department of the Environment and Energy, Canberra. Available from: https://biodiversity.org.au/afd/taxa/BUPRESTIDAE (accessed 11 March 2020).
Barker, S. (2006). 'Castiarina: Australia's richest jewel beetle genus'. South Australian Museum & University of Adelaide. 341 pp.
Bouchard, P., Smith, A.B.T., Douglas, H., Gimmel, M.L, Brunke, A.J. & Kanda, K. (2017). Biodiversity of Coleoptera. In: Footit, B. & Alder, P.H. (eds) 'Insect biodiversity: science and society.' Wiley-Blackwell, Oxford.
Yeates, D.K., Harvey, M.S. & Austin, A.D. (2003). New estimates for terrestrial arthropod species‐richness in Australia. Records of the South Australian Museum, Monograph Series 7: 231–241.
